On Air Now
The Capital Evening Show with Jimmy Hill 7pm - 10pm
9 November 2020, 12:24 | Updated: 9 November 2020, 12:29

The first Covid-19 vaccine is looking promising, exceeding the expectations of most scientists.
The first coronavirus vaccine can prevent more than 90 per cent of people from contracting Covid-19, large-scale trials by Pfizer/BioNTech have shown.
Boris Johnson Assures Lockdown Will Only Last Four Weeks
The analysis shows a better performance than most experts had hoped for, after regulators said they would approve a vaccine that is 50 per cent effective.

"Increasing evidence" shouting and singing increases coronavirus transmission
The company claim there have been no serious side-effects.
Their vaccine has been tested on 43,500 people in six countries.
“Today is a great day for science and humanity. The first set of results from our phase 3 Covid-19 vaccine trial provides the initial evidence of our vaccine’s ability to prevent Covid-19,” said Dr Albert Bourla, the Pfizer chairman and CEO.
“We are reaching this critical milestone in our vaccine development program at a time when the world needs it most with infection rates setting new records, hospitals nearing over-capacity and economies struggling to reopen.”
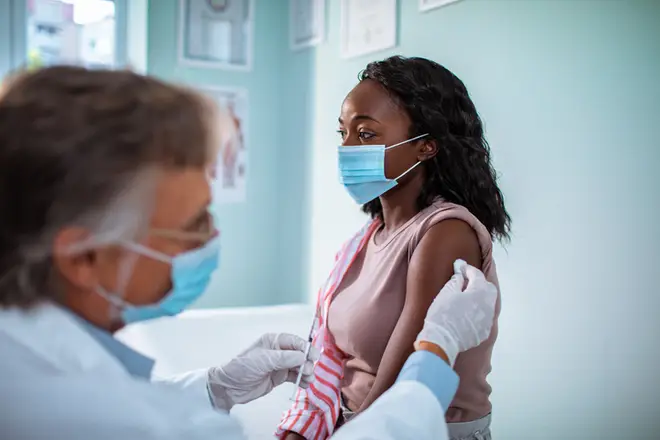
There are around 12 vaccine trials that have reached the final stages of testing, but this is the first to show any results.
The vaccine works by injecting part of the virus’ genetic code in order to train the immune system.
Two doses would be needed, three weeks apart.
If approved by regulators this month, Pfizer are aiming to supply 50 million doses by the end of 2020 and around 1.3 billion by the end of 2021.
The UK has already ordered 30 million doses.
> Download Our App For All The Latest News